MetallurgyHard
Question
Among the following groups of oxides, the group that cannot be reduced by carbon to give the respective metals.
Options
A.Cu2O, SnO2
B.Fe2O3, ZnO
C.CuO, K2O
D.PbO, FeO.
Solution
(A) Cu2O + C 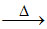 2Cu + CO ; SnO2 + 2C (anthracite)
2Cu + CO ; SnO2 + 2C (anthracite) 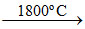 Sn + 2CO.
Sn + 2CO.
(B) 2Fe2O3 + 3C → 4Fe (spongy iron) + 3CO2 ; ZnO + C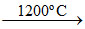 Zn + CO.
Zn + CO.
(C) Oxides of highly reactive metals (like K2O) are not reduced by carbon. K lie above hydrogen in electrochemical series and so it acts as strong reducing agent. K is obtained by electrolytic reduction of their fused salts.
(D) FeO + C → Fe + CO ; PbO + C → Pb + CO
(B) 2Fe2O3 + 3C → 4Fe (spongy iron) + 3CO2 ; ZnO + C
(C) Oxides of highly reactive metals (like K2O) are not reduced by carbon. K lie above hydrogen in electrochemical series and so it acts as strong reducing agent. K is obtained by electrolytic reduction of their fused salts.
(D) FeO + C → Fe + CO ; PbO + C → Pb + CO
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
Which of the following does not contain Mg:...In the electrolysis of alumina, cryolite is added to :...Carbon and CO gas are used to reduce which of the following pairs of metal oxides forexpiration of metals ?...Cyanide process is used for the extraction of...Match the ores listed in column I with the type of ores listed in column II and select the correct. alternate. Column I ...