ElectroMagnetic InductionHard
Question
A diatomic molecule is made of two masses m1and m2 which are separated by a distance r. Ifwe calculate its rotational energy by applyingBohr′s rule of angular momentum quantization,its energy will be given by : (n is an integer)
Options
A.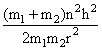
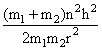
B.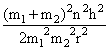
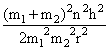
C.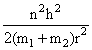
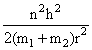
D.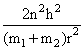
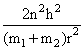
Solution
Energy 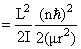
where μ = reduced mass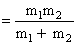
So Energy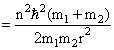
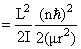
where μ = reduced mass
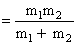
So Energy
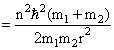
Create a free account to view solution
View Solution FreeMore ElectroMagnetic Induction Questions
Among the following properties describing diamagnetism identify the property that is wrongly stated :-...Two identical coaxial circular loops carry a current i each circulating in the same direction. If the loops approach eac...A square coil ACDE with its plane vertical is released from rest in a horizontal uniform magnetic field of length 2L. Th...A conducting rod PQ of length 5 m oriented as shown in figure is moving with velocity (2m /s) î without any rotatio...An inductance L and a resistance R are joined to a battery. After some time, battery is disconnected but L and R remains...