Analytical ChemistryHard
Question
The reagents, NH4Cl and aqueous NH3 will precipitate :
Options
A.Ca2+
B.Al3+
C.Mg2+
D.Zn2+
Solution
The KSP of Al(OH)3 is low as compared to that of the hydroxides of other metals. The OH- concentration provided by ammonium hydroxide in presence of NH4Cl is just sufficient to precipitate Al3+ as Al(OH)3.
Al3+ + NH3 + H2O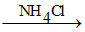 Al(OH)3 ↓ (gelatinous white) + NH4+.
Al(OH)3 ↓ (gelatinous white) + NH4+.
Al3+ + NH3 + H2O
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
The following can be used to regulate the concentration of OH- ions for the scheme of basic radical analysis (III group)...Hg2I2↓ (green) productsWhich of the following statement is correct with respect to the products ?...The composition of golden spangles is :...Consider the following statements, S1 : yellow precipitate of SrCrO4 is soluble in large quantity of water.S2 : yellow p...Select the correct statement with respect to Ca2+ ions....