Analytical ChemistryHard
Question
NH4SCN can be used to test one or more out of Fe3+, Co2+,Cu2+ :
Options
A.Fe3+ only
B.Co2+ , Cu2+ only
C.Fe3+ Cu2+ only
D.All
Solution
Fe3+ + 3SCN- → Fe(SCN)3 (deep red colouration)
Co2+ + 4SCN-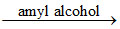 [Co(SCN)4]2- (blue colouration)
[Co(SCN)4]2- (blue colouration)
Cu2+ + 2SCN- → Cu(SCN)2 ↓(black) ; 2Cu(SCN)2 → 2CuSCN ↓(white) + (SCN)2.
Co2+ + 4SCN-
Cu2+ + 2SCN- → Cu(SCN)2 ↓(black) ; 2Cu(SCN)2 → 2CuSCN ↓(white) + (SCN)2.
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
Upon treatment with ammonical H2S , the metal ion that precipitates as a sulfide is -...The pair in which both species have iron is :...There are four test tubes containing dilute HCl, BaCl2, CdCl2 and KNO3 solutions. Which of the following reagents will h...A metal nitrate reacts with KI solution to give yellow precipitate which on addition of excess of more concentrated solu...Beilstein test is used for the detection of :...