Analytical ChemistryHard
Question
Fe(OH)3 and Cr(OH)3 precipitates can be completely separated by :
Options
A.Aq. NH3
B.HCl
C.NaOH/H2O2
D.H2SO4
Solution
Both dissolves in acids. Cr(OH)3 is partially soluble while Fe(OH)3 is not soluble in aqueous NH3. Only Cr(OH)3 not Fe(OH)3 is soluble by NaOH/H2O2 according to the reaction.
Cr(OH)3 ↓ (green)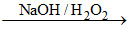 CrO42- (yellow solution) + H2O.
CrO42- (yellow solution) + H2O.
Cr(OH)3 ↓ (green)
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
Which one of the following imparts green colour to the burner flame ?...In which of the following solvents, AgBr will have the highest solubility ?...Which of the statements is not true ?...Which of the following statement is not correct ?...In fifth group, (NH4)2CO3 is added to precipitate out the carbonates. We do not add Na2CO3 because :...