Analytical ChemistryHard
Question
Consider the following observation :
Mn+ + HCl (dilute) → white precipitate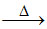 water soluble
water soluble 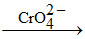 yellow precipitate.
yellow precipitate.
The metal ion Mn+ will be :
Mn+ + HCl (dilute) → white precipitate
The metal ion Mn+ will be :
Options
A.Hg2+
B. Ag+
C.Pb2+
D.Sn2+
Solution
Pb2+ + 2Cl- → PbCl2 ↓ (soluble in hot water) ; PbCl2 + CrO42- → PbCrO4 ↓ (yellow) + 2Cl-.
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
A black sulphide is formed by the action of H2S on :...In which of the following reactions, white precipitate is obtained as one of the reaction products ?...A metal salt solution forms a yellow precipitate with potassium chromate in acetic acid, a white precipitate with dilute...The following can be used to regulate the concentration of OH- ions for the scheme of basic radical analysis (III group)...An aqueous solution contains both Al3+ & Zn2+.To this solution NH4OH is added in excess....