s-Block ElementsHard
Question
Which of the following statement(s) is/are correct ?
Options
A.Sodium bicarbonate is more soluble than sodium carbonate.
B.Sodium hydroxide is used in the purification of bauxite.
C.Sodium bicarbonate is used as antacid and for making baking powder.
D.Potassium hydroxide is used in the manufacture of soft soaps.
Solution
(A) Sodium bicarbonate is sparingly soluble while sodium carbonate is fairly soluble in water.
(B) Al2O3 + 2NaOH → 2NaAlO2 (soluble) + H2O. (Bayer′s process).
(C) NaHCO3 + H2O ⇋ NaOH + H2CO3 - reduces acidity.
2NaHCO3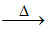 Na2CO3 + H2O + CO2 - CO2 liberated produces porosity and thus makes bread soft.
Na2CO3 + H2O + CO2 - CO2 liberated produces porosity and thus makes bread soft.
(D) One of the uses of potassium hydroxide is in making the soft soaps
(B) Al2O3 + 2NaOH → 2NaAlO2 (soluble) + H2O. (Bayer′s process).
(C) NaHCO3 + H2O ⇋ NaOH + H2CO3 - reduces acidity.
2NaHCO3
(D) One of the uses of potassium hydroxide is in making the soft soaps
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Sodium carbonate can be manufactured by Solvay′s process but potassium carbonate cannot be prepared because:...Be and Al have following resemblance due to diagonal relationship :...In certain matters, lithium differs from other alkali metals, the main reason for this is :...Which one of the following electrolyte is used in Down′s process of extracting sodium metal ?...The pair (s) of compounds which can exist together in aqueous solutions is/are ?...