s-Block ElementsHard
Question
Compounds of alkaline earth metals are less soluble in water than the corresponding alkali metal salts due to
Options
A.their high ionisation energy
B.their low electronegativity
C.their low hydration energy
D.their high lattice energy
Solution
Along the period, atomic size decreases and nuclear charge increases and thus lattice energy increases,
lattice energy ∞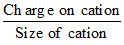
lattice energy ∞
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Slaked lime, Ca(OH)2 is used :...Sodium has, as compared to potassium :...When magnesium burns in air, compounds of magnesium formed are magnesium oxide and :...Select the correct statement with respect to the deep blue solution of an alkali metal in liquid ammonia....Alkali metals possess metallic lustre when freshly cut because :...