s-Block ElementsHard
Question
In which of the following pairs the two species are not isostructural ?
Options
A.AlF63- and SF6
B.CO32- and NO3-
C.PCl4+ and SiCl4
D.PF5 and BrF5
Solution
(1) AlF63- and SF6 → both are octahedral.
(2) CO32- and NO3 → both are triangular planar
(3) PCl4+ and SiCl4 → both are tetrahedral
(4) PF5 and BrF5 → PF5 has 5bp, 0 lp (sp3d)Structure is trigonal bipyramidal
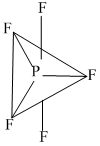
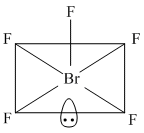
BrF5 has 5bp & 1 lp (sp3d2) and structure issquare pyramidal
(2) CO32- and NO3 → both are triangular planar
(3) PCl4+ and SiCl4 → both are tetrahedral
(4) PF5 and BrF5 → PF5 has 5bp, 0 lp (sp3d)Structure is trigonal bipyramidal

BrF5 has 5bp & 1 lp (sp3d2) and structure issquare pyramidal

Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
The pairs of compound which cannot exist together in aqueous solution are :...Which one of the following alkaline earth metal sulphates has its hydration enthalpy greater than its lattice enthalpy ?...Lithium nitrate on heating gives :...Largest difference radii is found in case of the pair...The principal products obtained on heating iodine with concentrated caustic soda solution is :...