Magnetic field due to currentHard
Question
The equilibrium constant (KC) for the reaction N2(g) + O2(g) → 2NO(g) at temperature T is 4 × 10-4. The value of KC for the reaction. NO(g)→ 1/2 N2 (g) + 1/2 O2(g) at the sametemperature is :-
Options
A.50.0
B.0.02
C.2.5 × 102
D.4 × 10-4
Solution
N2 + O2 → 2NO KC = 4 × 10-4
2NO → N2 + O2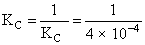
NO →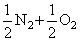
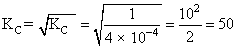
2NO → N2 + O2
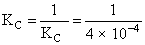
NO →
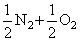
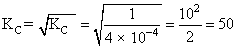
Create a free account to view solution
View Solution FreeMore Magnetic field due to current Questions
Which among the following will be named as dibromidobis (ethylene diamine) chromium (III) bromide ?...The dimensional formula for the physical quantity is (E = electric field and B = magnetic field)...The magnetic field lines due to bar magnetic are correcticle shown in...A helium nucleus is moving in a circular path of radius 0.8m. If it takes 2 sec to complete one revolution. Find out mag...A uniform beam of positively charged particles is moving with a constant velocity parallel to another beam of negatively...