s-Block ElementsHard
Question
Consider the following statements ;
S1 : Mg2+ ion is bigger than Li+.
S2 : LiNO3 and NaNO3 both on heating give NO2 and dioxygen.
S3 : All alkali metal hydrogencarbonates exist in solid state.
S4 : All alkali metal halides form hydrates.
and arrange in the order of true/false.
S1 : Mg2+ ion is bigger than Li+.
S2 : LiNO3 and NaNO3 both on heating give NO2 and dioxygen.
S3 : All alkali metal hydrogencarbonates exist in solid state.
S4 : All alkali metal halides form hydrates.
and arrange in the order of true/false.
Options
A.T T F F
B.T T T F
C.F F F F
D.T F F F
Solution
S1 : Both are diagonally related ; because of more positive charge on Mg, Mg2+ is smaller than Li+.
Li+ = 76 pm , Mg2+ = 72 pm.
S2 : 4 LiNO3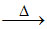 2 Li2O + 4 NO2 + O2 ; 2 NaNO3
2 Li2O + 4 NO2 + O2 ; 2 NaNO3 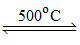 2 NaNO2 + O2 ;
2 NaNO2 + O2 ;
4NaNO3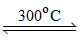 2Na2O + 5 O2 + 2 N2
2Na2O + 5 O2 + 2 N2
S3 : Lithium is least basic ; so LiHCO3 is thermally unstable and is not found in solid state.
S4 : Except Li+, due to bigger size of ions they have low hydration enthalpies. Hence except lithium, all alkali metal halides do not form hydrates.
Li+ = 76 pm , Mg2+ = 72 pm.
S2 : 4 LiNO3
4NaNO3
S3 : Lithium is least basic ; so LiHCO3 is thermally unstable and is not found in solid state.
S4 : Except Li+, due to bigger size of ions they have low hydration enthalpies. Hence except lithium, all alkali metal halides do not form hydrates.
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Sodium carbonate can be manufactured by Solvay′s process but potassium carbonate cannot be prepared because:...Setting of plaster of paris involves :...A fire work gave bright crimson light. It probably contained a salt of :...Peroxide ion is present in :...Which of the following is different from other three oxides ?...