s-Block ElementsHard
Question
Select the correct statement with respect to sodium peroxide ?
Options
A.It decolourises the acidifed KMnO4 solution.
B.On heating with oxygen at 450oC and 300 atm pressure, it becomes paramagnetic.
C.It is obtained along with sodium metal, when sodium oxide is heated to a temperature more than 400oC.
D.It gives both hydrogen peroxide and oxygen gas with water as well as with sulphuric acid.
Solution
(A) 2MnO4- (pink) + 16H+ + 5 O22- → 2Mn2+ + 8 H2O + 5O2.
(B) Na2O2 + O2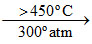 2NaO2 (O2- contains one unpaired electron in π* MO).
2NaO2 (O2- contains one unpaired electron in π* MO).
(C) 2 Na2O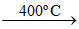 Na2O2 + 2 Na.
Na2O2 + 2 Na.
(D) gives only H2O2.
(B) Na2O2 + O2
(C) 2 Na2O
(D) gives only H2O2.
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Na2[Be(OH)4] is formed when ;...Which of the following oxides is formed when potassium metal is burnt in excess air ?...A piece of magnesium ribbon was heated to redness in an atmosphere of nitrogen and on cooling, water was added, the gas ...Which is/are not correct configuration of s-block elements :...Chemical (A) is used for water softening to remove temporary hardness. A reacts with Na2 CO3 to generate caustic soda. W...