p-Block elementsHard
Question
F2 is formed by reacting K2MnF6 with :
Options
A.SbF5
B.MnF3
C.KSbF6
D.MnF5
Solution
K2[MnF6] + 2SbF5 → 2K[SbF6] + MnF3 + 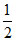 F2
F2
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
Ca + C2 → CaC2 $\overset{\quad N_{2}\quad}{\rightarrow}$ ACompound (A) is used as a/an...Which of the following reactions depict the oxidising behaviour of H2SO4 ?...The statement true for N3- is :...Which of the following statements regarding ortho boric acid (H3BO3) is false ?...The formation of O2+ [PtF6]- is the basis for the formation of xenon fluorides. This is because :...