Magnetic field due to currentHard
Question
Ortho-Nitrophenol is less soluble in water than p- and m- Nitrophenols because :-
Options
A.Melting point of o-Nitrophenol is lower than those of m- and p- isomers
B.o-Nitrophenol is more volatile in steam than those of m- and p- isomers
C.o-Nitrophenol shows Intramolecular H-bonding
D.o-Nitrophenol shows Intermolecular H-bonding
Solution
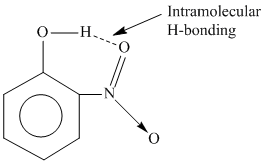
due to presence of intramolecular H-bonding its interaction with water molecule will be poor, resulting into less solubility of the compound. While para & meta derivative forms intermolecular H-bonding with water.
Create a free account to view solution
View Solution FreeMore Magnetic field due to current Questions
An ionised gas contains both positive and negative ions. If it is subjected simultaneously to an electric field along th...A magnetic field exists in the region a , in the region 2a 0 is a positive constant. A positive point charge moving with...A particle of charge ′q′and mass ′m′ enters normally (at point P) in a region of magnetic field ...A long insulated copper wire is closely wound as a spiral of ′N′ turns. The spiral has inner radius ′a...Needles N1, N2 and N3 are made of a ferromagnetic, a paramagnetic and a diamagnetic substance respectively. A magnet whe...