p-Block elementsHard
Question
Which of the following statement is false?
Options
A.An acidified solution of titanium salt gives yellow or orange colour with H2O2.
B.An acidified solution of K2Cr2O7 gives a bright blue coloured compound with H2O2 which is soluble in ether.
C.Reaction of HCHO with H2O2 in presence of pyrogallol in alkaline solution liberates hydrogen gas.
D.Anhydrous H2O2 is more stable and is kept in glass bottles.
Solution
(A) An acidified solution of titanium salt gives yellow or orange colour with H2O2.
Ti+4 + H2O2 + 2H2O → H2TiO4 (yellow/orange red) + 4H+
(B) Cr2O72- + 2H+ + 4H2O2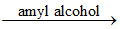 2CrO5 + 5H2O
2CrO5 + 5H2O
CrO5 bright blue coloured compound soluble in diethyl ether, amyl alcohol and amyl acetate.
(C) 2HCHO + H2O2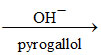 2HCOOH + H2
2HCOOH + H2
(D) Its aqueous solution is more stable than the anhydrous liquid where it decomposes into water and O2 slowly on exposure to light.
2H2O2 → 2H2O + O2
H2O2 is not kept in glass containers because traces of alkali metal ions from the glass can catalyse the explosive decomposition of H2O2.
Ti+4 + H2O2 + 2H2O → H2TiO4 (yellow/orange red) + 4H+
(B) Cr2O72- + 2H+ + 4H2O2
CrO5 bright blue coloured compound soluble in diethyl ether, amyl alcohol and amyl acetate.
(C) 2HCHO + H2O2
(D) Its aqueous solution is more stable than the anhydrous liquid where it decomposes into water and O2 slowly on exposure to light.
2H2O2 → 2H2O + O2
H2O2 is not kept in glass containers because traces of alkali metal ions from the glass can catalyse the explosive decomposition of H2O2.
Create a free account to view solution
View Solution Free