p-Block elementsHard
Question
Select the correct statement.
Options
A.Ostwald′s method of preparation of HNO3 is based upon catalytic oxidation of NH3 by atmospheric oxygen.
B.HNO2 acts as both oxidising and reducing agents.
C.NO2 reacts with O3 to form N2O5.
D.All of these.
Solution
(A) 4NH3 + 5O2 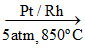 4NO (g) + 6H2O (g) ; principal reaction of Ostwald′s method of HNO3 manufacturing.
4NO (g) + 6H2O (g) ; principal reaction of Ostwald′s method of HNO3 manufacturing.
(B) HNO2 is weak oxidising agent and oxidises Fe2+ to Fe3+ and I- to I2 and it self reduced to N2O or NO. In contrary HNO2 is oxidised by KMnO4 and Cl2 forming NO3-.
(C) 2NO2 + O3 → N2O5 + O2
(B) HNO2 is weak oxidising agent and oxidises Fe2+ to Fe3+ and I- to I2 and it self reduced to N2O or NO. In contrary HNO2 is oxidised by KMnO4 and Cl2 forming NO3-.
(C) 2NO2 + O3 → N2O5 + O2
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
The pair of amphoteric hydroxides is...Which among the following factors is the most important in making fluorine the strongest oxidizing halogen?...he following acids have been arranged in the order of decreasing acid strength. Identify the correct order ClOH (I), BrO...Identify the incorrect statement with respect to ozone....Oxygen is more electronegative than sulphur, yet H2O is neutral. This is because...