Momentum and CollisionHard
Question
The pH of a 0.1 molar solution of the acid HQ is 3. The value of the ionization constant, Ka of this acid is :-
Options
A.1 × 10 -7
B.3 × 10 -7
C.1 × 10 -3
D.1 × 10 -5
Solution
pH = -log[H+] pH = 3
[H+] = 10-pH = 10-3
For weak acid
[H+] =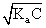
[H+]2 = Ka × C
(10-3)+ = Ka × 0.1
Ka = 10-5
[H+] = 10-pH = 10-3
For weak acid
[H+] =
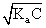
[H+]2 = Ka × C
(10-3)+ = Ka × 0.1
Ka = 10-5
Create a free account to view solution
View Solution FreeMore Momentum and Collision Questions
A liquid drop of diameter D breaks into 27 tiny drops. The resultant change in energy is : -...A vessel with a hole in its bottom is fastened on a cart. The mass of the vessel and the cart is M and the area of the v...The rate of flow of liquid through a capillary tube, in an experiment to determine the viscosity of the liquid, increase...If a water drop is kept between two glass plates, then its shape is : -...A jar filled with two non mixing liquids 1 and 2 having densities ρ1 and ρ2 respectively. A solid ball, made o...