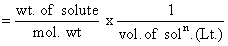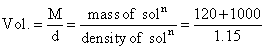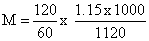Momentum and CollisionHard
Question
The density of a solution prepared by dissolving 120 g of urea (mol. mass = 60 u) in 1000 g of water is 1.15 g/mL. The molarity of this solution is :-
Options
A.2.05 M
B.0.50 M
C.1.78 M
D.1.02 M
More Momentum and Collision Questions
Pressures inside two soap bubbles are 1.01 and 1.02 atmospheres. Ratio between their volumes is : -...Four point masses, of a value m are placed at the corners of a square ABCD of side l.The moment of interia of this syste...A cylindrical vessel filled with water is released on an inclined surface of angle θ as shown in figure. The fricti...Two men ′A′ and ′B′ are standing on a plank. ′B′ is at the middle of the plank and &...A particle moving with kinetic energy = 3J makes an elastic head - on collision with a stationary particle which has twi...