p-Block elementsHard
Question
Which is / are the correct statement(s) ?
Options
A.BeF2 readily coordinates two additional F- ions forming the [BeF4]2- complex.
B.One mole of borax in aqueous solution reacts with two moles of acid.
C.HCOONa as well as solid K3[Fe(CN)6] both on heating with concentrated sulphuric acid evolve carbon monoxide gas.
D.Carbon mono oxide when passed through a solution of iodine pentaoxide, I2O5 liberates iodine and carbon dioxide gases.
Solution
(A) [BeF4]2-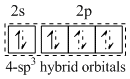 Four electron pairs - two form covalent and two form coordinate bonds.
Four electron pairs - two form covalent and two form coordinate bonds.
Two F- ions each donate an electron pair in to an empty sp3 hybrid orbital forming two coordinate bonds.
(B) [B4O5(OH)4]2- + 5H2O ⇋ 2B(OH)3 + 2 [B(OH)4]-
2[B(OH)4] + 2H3O+ → 2B(OH)3 + 4H2O
Only [B(OH)4]- formed in water reacts with HCl.
(C) HCOONa + H2SO4 → CO ↑ + Na+ + HSO4- + H2O.
K3[Fe(CN)6] + 6H2SO4 + 6H2O → 6CO + Fe3+ + 3 K+ + 6NH4+ + 6SO42-
Solid potassium ferrocyanide also liberates CO (g) on heating with concentrated H2SO4.
(D) 2I2O5 + 5CO → 5CO2 + 2I2
 Four electron pairs - two form covalent and two form coordinate bonds.
Four electron pairs - two form covalent and two form coordinate bonds. Two F- ions each donate an electron pair in to an empty sp3 hybrid orbital forming two coordinate bonds.
(B) [B4O5(OH)4]2- + 5H2O ⇋ 2B(OH)3 + 2 [B(OH)4]-
2[B(OH)4] + 2H3O+ → 2B(OH)3 + 4H2O
Only [B(OH)4]- formed in water reacts with HCl.
(C) HCOONa + H2SO4 → CO ↑ + Na+ + HSO4- + H2O.
K3[Fe(CN)6] + 6H2SO4 + 6H2O → 6CO + Fe3+ + 3 K+ + 6NH4+ + 6SO42-
Solid potassium ferrocyanide also liberates CO (g) on heating with concentrated H2SO4.
(D) 2I2O5 + 5CO → 5CO2 + 2I2
Create a free account to view solution
View Solution Free