p-Block elementsHard
Question
Carbon monoxide is prepared by :
Options
A.heating formic acid with conc. H2SO4
B.heating potassium ferrocyanide with conc H2SO4
C.heating malonic acid with P4O10
D.hydrolysis of Mg2C3
Solution
HCOOH 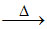 H2O + CO ; K4[Fe(CN)6] + 6H2SO4 + 6H2O → 2K2SO4 + FeSO4 + 3(NH4)2SO4 + 6CO
H2O + CO ; K4[Fe(CN)6] + 6H2SO4 + 6H2O → 2K2SO4 + FeSO4 + 3(NH4)2SO4 + 6CO
Malonic acid gives C3O2 and Mg2C3 gives 1-propyne.
Malonic acid gives C3O2 and Mg2C3 gives 1-propyne.
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
In the preparation of HBr or HI , NaX (X = Br, I) is treated with H3PO4 and not by concentrated H2SO4 since,...When ammonia is oxidsed by oxygen in the presence of platinum at 800oC, the gas obtained is :...Sodium thiosulphate is prepared by :...Borax is:...A complex cross-linked polymer (silicone) is formed by...