Ionic EquilibriumHard
Question
Which of the following is incorrect statement.
Options
A.If K1 and K2 for dissociation of H2A is 10-7 and 10-14 then A2- concentration in 0.1 M H2A solution is 10-7
B.A solution of pH = 1 has H+ ion concentration 3 times than that of a solution of pH = 3
C.If a salt solution of weak acid & weak base is diluted upto limited extent then its pH will decrease
D.pH of acidic buffer solution depends upon pKa and relative molar amount of weak acid and its conjugate base.
Solution
[A2-] = Ka2 = 10-5
pH = 1 pH = 3
[H+]1 = 10-1 [H+]2 = 10-3
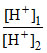 = 100
= 100
pH =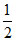 (pKw + pKa - pKb)
(pKw + pKa - pKb)
pH is independent of dilution with in a limit since no concentration term in pH expression.
pH = 1 pH = 3
[H+]1 = 10-1 [H+]2 = 10-3
pH =
pH is independent of dilution with in a limit since no concentration term in pH expression.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A 0.1 mole of AgNO3 is dissolved in 1 L of 1 M-NH3. If 0.01 mole of NaCl is added to this solution, will AgCl(s) precipi...A 40.0 ml solution of weak base, BOH is titrated with 0.1 N – HCl solution. The pH of the solution is found to be 10.0 a...Which shows the maximum rate of alkaline hydrolysis :-...Incorrect order of ionic character :...Which of the following mixture gives a buffer solution with $pH = 9.25$ ?Given : ${pK}_{b}\left( {NH}_{4}OH \right) = 4....