Solid StateHard
Question
AB crystallizes in a body centered cubic lattice with edge length ′a′ equal to 387 pm. The distance between two oppositely charged ions in the lattice is
Options
A.335 pm
B.250 pm
C.200 pm
D.300 pm
Solution
For body centered cubic (bcc) lattice,
distance between two positively charged ions,
d =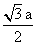
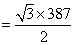 pm
pm
= 335.15 pm
distance between two positively charged ions,
d =
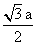
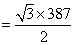 pm
pm= 335.15 pm
Create a free account to view solution
View Solution FreeMore Solid State Questions
Frenkel defect is observed in...In a sodium chloride crystal, the unit cell edge length is a. The option(s) representing the correct combination is/areO...The density of KCl is 1.9893 g-cm-3 and the length of a side of unit cell is 6.29082 A0. The value of Avogadro′s n...In a hypothetical solid, ‘C’ atoms form CCP lattice. ‘A’ atoms occupy all tetrahedral voids and ‘B’ atoms occupy all oct...NaCl shows Schottky defects and AgCl Frenkel defects. Their electrical conductivity is due to :...