IsomerismHard
Question
In the following the most stable conformation of n-butane is
Options
A.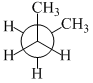

B.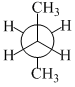

C.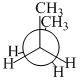

D.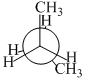

Solution
The conformation in which the heavier groups are present at maximum possible distance, so that the force of repulsion get weak, is more stable.
Among the given conformations of n-butane, the conformation shown in option (b) ie, anti conformation is most stable as in it the bulkier group (ie, CH3 group) are preasent at maximum possible distance.
Among the given conformations of n-butane, the conformation shown in option (b) ie, anti conformation is most stable as in it the bulkier group (ie, CH3 group) are preasent at maximum possible distance.
Create a free account to view solution
View Solution FreeMore Isomerism Questions
What is true for 1,2 − pentadiene −...Which will show geometrical isomerism ?...Possible number of disubstituted benzene isomers is -...Increasing order of stability among the three main conformation (i.e. eclipse, anti, gauche) of ethylene glycol is :...But-2-ene exhibits cis-trans-isomerism due to-...