Nitrogen Containing CompoundHard
Question
Which of the following statement about primary amine is false ?
Options
A.Alkyl amines are stronger bases than aryl amines
B.Alkyl amines reacts with nitrous acid to produce alcohols
C.Alkyl amines reacts with nitrous acid to produced phenols
D.Alkyl amines are stronger bases than ammonia
Solution
(i) Presence of electron withdrawing substituent deceases the basicity while the presence of electron releasing substituent like, - CH3, - C2H5 etc increases the acidity.
(ii) HNO2 convert - NH2 group of aliphatic amine into - OH while that of aromatic amines into - NNCl.
Since, phenyl group is a electron withdrawing group, it decreases the basicity. Alkyl group, on the other hand, being electron releasing, increases the basicity. Thus, alkyl amines are more basic as compared to aryl amines as well as ammonia.

Thus, HNO2 (nitrous acid) converts alkyl amines to alcohols.
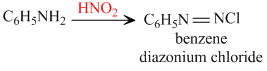
Thus, HNO2 does not convert aryl amines into phenol.
(ii) HNO2 convert - NH2 group of aliphatic amine into - OH while that of aromatic amines into - NNCl.
Since, phenyl group is a electron withdrawing group, it decreases the basicity. Alkyl group, on the other hand, being electron releasing, increases the basicity. Thus, alkyl amines are more basic as compared to aryl amines as well as ammonia.

Thus, HNO2 (nitrous acid) converts alkyl amines to alcohols.
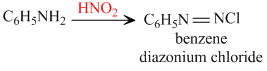
Thus, HNO2 does not convert aryl amines into phenol.
Create a free account to view solution
View Solution Free