General Organic ChemistryHard
Question
The correct order of increasing reactivity of C - X bond towards nuceophile in the following compounds is
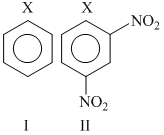
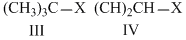

Options
A.I < II < IV < III
B.II < III < I < IV
C.IV < III < I < II
D.III < II < I < IV
Solution
Alkyl halides are more reactive towards nucleophilic substitution of carbocation intermediate formed.
Among the given halides, aryl halides (C6H5X) is least reactive towards nucleophile as in it the C - X bond acquire some double bond character due to resonance. Presence of electron withdrawing groups like - NO2 at ortho and para positions facilitate the nucleophilc displacement of - X of aryl halides. Among alkyl halides, 3o halides are more reactive as compared to 2o halides due to the formation of more stable carbocation. Hence, the order of reactivity of C - X bond towards nucleophlie is as
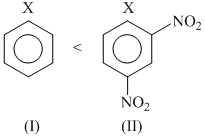
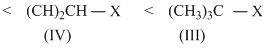
Among the given halides, aryl halides (C6H5X) is least reactive towards nucleophile as in it the C - X bond acquire some double bond character due to resonance. Presence of electron withdrawing groups like - NO2 at ortho and para positions facilitate the nucleophilc displacement of - X of aryl halides. Among alkyl halides, 3o halides are more reactive as compared to 2o halides due to the formation of more stable carbocation. Hence, the order of reactivity of C - X bond towards nucleophlie is as

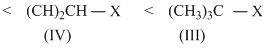
Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
The geometrical isomerism is shown by...Arrange the Stability of following I II III...Turpentine oil can be purified by :...Consider the following three halides -(a) CH3 − CH2 − Cl (b) CH2 = CH − Cl (c) C6H5 − ClArrange ...The IUPAC name of the compound having the formula is...