General Organic ChemistryHard
Question
Which one of the following compounds has the most acidic nature ?
Options
A.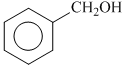

B.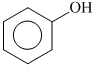

C.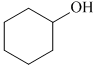

D.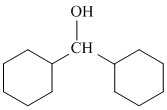

Solution
Presence of electron withdrawing substituent increases the acidity while electron releasing substituent, when present, decreases the acidity.
Phenyl is an electron withdrawing substituent while -CH3 is an electron releasing substituent. Moreover, phenoxide ion is more resonance stabilised as compared to benzyloxide ion, thus releases proton more easily. That′s why is a strong acid among the given.
Phenyl is an electron withdrawing substituent while -CH3 is an electron releasing substituent. Moreover, phenoxide ion is more resonance stabilised as compared to benzyloxide ion, thus releases proton more easily. That′s why is a strong acid among the given.
Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
Which of the following hydrocarbon groups designated as I, II, III, IV and V has planar geometry -Phenyl Cyclohexyl Cycl...CH3Br + Nu- → CH3 - Nu + Br- The decreasing order of the rate of the above reaction with nucleophiles (Nu-) A to D...Which among the following species is an ambident nucleophile :...Carbocations may be stabilised by -...In the replacement reaction The reaction will be most favourable if M happens to be:...