Gaseous StateHard
Question
The kinetic energy of N molecules of O2 is x joule at - 123oC. Another sample of O2 at 27oC has a kinetic energy of 2 x. The latter sample contains ____ molecules of O2.
Options
A.N
B.N/2
C.2N
D.3N
Solution
K.EO2 = 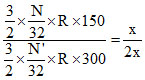 ⇒ K.EO2 =
⇒ K.EO2 = 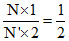
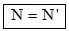 Therefore, (A) option is correct.
Therefore, (A) option is correct.
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
A hypothetical gaseous element having molecular formula Mx may be changed to another gaseous allotrope having molecular ...The compressibility of a gas is less than unity at STP. Therefore,...The product of PV is plotted against P at two temperatures T1 and T2 and the ′result is shown in figure. What is c...The volume of CO2 produced by the combustion of 40 ml of gaseous acetone in excess of oxygen is...For the non-zero values of force of attraction between gas molecules, gas equation will be :...