SHMHard
Question
The pair(s) of coordination complex/ion exhibiting the same kind of isomerism is(are) -
Options
A.[Cr(NH3)5Cl]Cl2 and [Cr(NH3)4Cl2]Cl
B.[Co(NH3)4Cl2]+ and [Pt(NH3)2(H2O)Cl]+
C.[CoBr2Cl2]2- and [PtBr2Cl2]2-
D.[Pt(NH3)3(NO3)] Cl and [Pt(NH3)3Cl] Br
Solution
(A) [Cr(NH3)5Cl]Cl2 does not show any isomerism while [Cr(NH3)4Cl2]Cl shows geometrical isomerism
(B) [Co(NH3)4Cl2]+ and [Pt(NH3)2(H2O) (Cl)]+
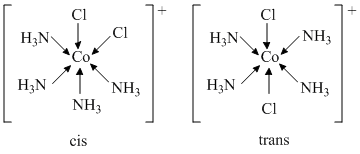
hence, [Co(NH3)4Cl2]+ ion shows geometrical isomerism.
[Pt(NH3)2(H2O) (Cl)]+
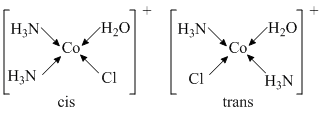
hence, [Pt(NH3)2(H2O)(Cl)]+ ion shows geometrical isomerism
(C) [CoBr2Cl2]2- and [PtBr2Cl2]2-
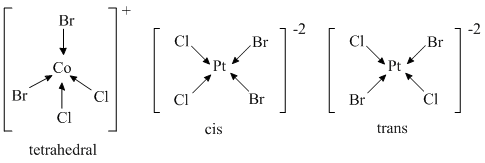
does not show any isomerism square planar and shows geometrical isomerism
(D) [Pt(NH3)3(NO3)]Cl and [Pt(NH3)3Cl]Br
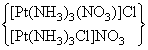 show ionisation isomerism
show ionisation isomerism
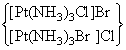 show ionisation isomerism
show ionisation isomerism
hence, Ans is (B, D)
(B) [Co(NH3)4Cl2]+ and [Pt(NH3)2(H2O) (Cl)]+

hence, [Co(NH3)4Cl2]+ ion shows geometrical isomerism.
[Pt(NH3)2(H2O) (Cl)]+
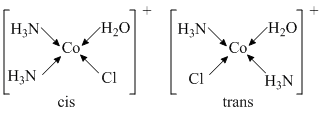
hence, [Pt(NH3)2(H2O)(Cl)]+ ion shows geometrical isomerism
(C) [CoBr2Cl2]2- and [PtBr2Cl2]2-

does not show any isomerism square planar and shows geometrical isomerism
(D) [Pt(NH3)3(NO3)]Cl and [Pt(NH3)3Cl]Br
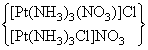 show ionisation isomerism
show ionisation isomerism 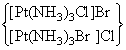 show ionisation isomerism
show ionisation isomerismhence, Ans is (B, D)
Create a free account to view solution
View Solution FreeMore SHM Questions
The spring constant of a spring is K. When it is divided into n equal parts, then what is the spring constant of one pie...A particle execute simple harmonic motion between x = - A and x = + A. he time taken for it go to O to is T1 and go from...The equation of a simple harmonic wave is given byy = 3 sin (50t - x) where x and y are in metres and t is in seconds. T...In fig.1 if the time period is T then calculate time period for fig.2 in which two springs are connected in series with ...An oscillating pendulum stops, because its energy...