Gaseous StateHard
Question
A gaseous mixture of three gases A, B and C has a pressure of 10 atm. The total number of moles of all the gases is 10. If the partial pressure of A and B are 3.0 and 1.0 atm respectively and if C has mol. wt. of 2.0, what is the weight of C in g present in the mixture ?
Options
A.6
B.8
C.12
D.3
Solution
Pressure of Total mixture = 10 atm
PA + PB + PC = 10
3 + 1 + PC = 10 ⇒ PC = 6 atm
Total moles of mixture = 10
nA + nB + nC = 10
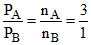 ⇒
⇒ 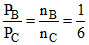
Let nA = K ⇒ nB =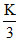 nc =
nc = 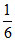 nB = 2K
nB = 2K
⇒ K +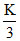 + 2K = 10 ⇒
+ 2K = 10 ⇒ 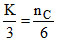 ⇒ nC = 2K
⇒ nC = 2K
⇒ K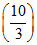 = 10 K = 3, ⇒ nA = 3
= 10 K = 3, ⇒ nA = 3
nB = 1
nC = 6
∴ weight of ′C′ in mixture = 2 × 6 = 12
PA + PB + PC = 10
3 + 1 + PC = 10 ⇒ PC = 6 atm
Total moles of mixture = 10
nA + nB + nC = 10
Let nA = K ⇒ nB =
⇒ K +
⇒ K
nB = 1
nC = 6
∴ weight of ′C′ in mixture = 2 × 6 = 12
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
At what temperature will the total KE of 0.3 mol of He be the same as the total KE of 0.40 mol of Ar at 400 K ?...Identify the correct statements when a fixed amount ideal gas is heated in from 300 to 600 K in a container fitted with ...The average velocity of an ideal gas molecule at 27oC is 0.3/s. The avarage velocity at 927oC will be :...A gas cylinder containing cooking gas can withstand a pressure of 14.9 atmosphere. The pressure guaze of cylinder indica...Equal weight of SO2 and O2 are mixed in an container at 25oC. The fraction of the total pressure exerted by O2 is :-...