ElectrochemistryHard
Question
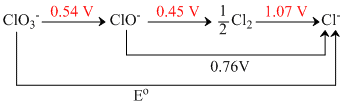
The Eo in the given figure is,
Options
A.0.5
B.0.6
C.0.7
D.0.8
Solution
ClO3- + 2H2O + 4e → ClO- + 4OH- ; ᐃG1o
ClO- + H2O + e →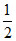 Cl2 + 2OH- ; ᐃG2o
Cl2 + 2OH- ; ᐃG2o
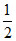 Cl2 + e → Cl- ; ᐃG3o
Cl2 + e → Cl- ; ᐃG3o
__________________________________
ClO3- + 3H2O + 3e → Cl- + 6OH- ; ᐃGo
__________________________________
∴ ᐃGo = ᐃG1o + ᐃG2o + ᐃG3o
- 6FEo = - 4F × 0.54 - 1F × 0.45 - 1F × 1.07
Eo = +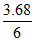 = + 0.61 V
= + 0.61 V
ClO- + H2O + e →
__________________________________
ClO3- + 3H2O + 3e → Cl- + 6OH- ; ᐃGo
__________________________________
∴ ᐃGo = ᐃG1o + ᐃG2o + ᐃG3o
- 6FEo = - 4F × 0.54 - 1F × 0.45 - 1F × 1.07
Eo = +
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The standard potentials of MnO4−|Mn2+ and MnO2|Mn2+ electrodes in acid solution are 1.51 and 1.23 V, respectively. Stand...EMF of the cell: Cd(s)|CdCl2.5H2O (sat.)|AgCl(s)|Ag(s) is +0.70 V at 0°C and +0.60 V at 50°C. If ΔHo and ΔSo are tempera...The standard reduction potential at 25°C of the reaction 2H2O + 2e− $\rightleftharpoons$H2 + 2OH− is −0.84 V. Calculate ...The standard potentials of OCl−/Cl− and Cl−/Cl2 are 0.94 V and −1.36 V, respectively. The Eo value of OCl−/Cl2 will be...When 12,000 coulombs of electricity is passed through the electrolyte, 3.0 g of a metal of atomic mass 96.5 g/mol is dep...