ElectrochemistryHard
Question
The reduction potential of hydrogen electrode when placed in a buffer solution is found to be - 0.413V. The pH of the buffer is -
Options
A.10
B.4
C.7
D.12
Solution
- 0.413 = 0 - 0 .059 log 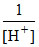 or
or 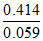 = - log H+ = pH or pH = 7
= - log H+ = pH or pH = 7
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
How many electrons are delivered at the cathode during electrolysis by a current of 1amp in 60 seconds?...Which one of the following statements is incorrect regarding an electrochemical cell?...Select the correct option if it is known that Ksp (AgCl) > Ksp (AgBr) > Ksp (AgI)...A gas ‘X’ at 1 atm is bubbled through a solution containing a mixture of 1 M – Y− and 1 M – Z− at 25°C. If the reduction...For a reaction A(s) + 2B+ → A2+ + B(s); KC has been found to be 1012. The EMF of the cell is...