Atomic StructureHard
Question
Change in angular momentum when an electron makes a transition corresponding to the 3rd line of the Balmer series in Li2+ ion is
Options
A.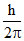
B.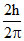
C.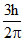
D.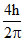
Solution
Change is angular momentum =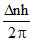 = (5 - 2)
= (5 - 2) 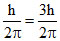
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Electromagnetic radiations of wavelength 242 nm is just sufficient to ionise Sodium atom. Then the ionisation energy of ...What possibly can be the ratio of the de Broglie wavelengths for two electrons each having zero initial energy and accel...The penultimate and outermost orbit of an element contains 10 and 2 electrons, respectively. If the outermost orbit is f...The correct set of four quantum numbers for the unpaired electron of chloride atom is...Total number of lone pair of electrons in I3- ion is...