Atomic StructureHard
Question
If first ionization potential of an atom is 16 V, then the first excitation potential will be :
Options
A.10.2 V
B.12 V
C.14 V
D.16 V
Solution
IP = 13.6Z2 = 16 (given).
1st excitation potential = 13.6 ×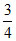 × Z2 = 16 ×
× Z2 = 16 × 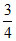 = 12 V.
= 12 V.
IP = 13.6Z2 = 16 (given).
1st excitation potential = 13.6 ×
IP = 13.6Z2 = 16 (given).
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The correct order of magnetic moments (spin only values in B.M.) among is...For a d-electron, the orbital angular momentum is :...Which of the following statements is/are correct for an electron of quantum numbers n = 4 and m = 2?...The potential energy of the electron present in the ground state of Be3+ ion is represented by:...Correct set of forur quantum numbers for the valence (outermost) electron of rubidium (Z = 37) is :...