NEETAtomic StructureHard
Question
The quantum numbers + 1/2 and - 1/2 for the electron spin represent -
Options
A.Rotation of the electron in clockwise and anticlockwise direction respectively.
B.Rotation of the electron in anticlockwise and clockwise direction respectively.
C.Magnetic moment of the electron pointing up and down respectively,
D.Two quantum mechanical spin states which have no classical analogue.
Solution
Spin quantum number does not comes from Schrodinger equation.
s = +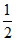 and -
and - 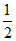 have been assigned arbitrarily.
have been assigned arbitrarily.
s = +
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
A green bulb and a red bulb are emitting the radiations with equal power. The correct relation between numbers of photon...The difference in angular momentum associated with the electron in two successive orbits of hydrogen atom in :-...The wavelength of first line of Lyman series of H-atom is 1216 Å. What will be the wavelength of first line of Lyman ser...The correct set of four quantum numbers for the valence electron of rubidium atom (Z = 37) is...Imagine an atom made up of a stationary proton and a hypothetical particle of double the mass of electron but having the...