ElectrostaticsHard
Question
Which of the following arrangements does not represent the correct order of the property stated against it ?
Options
A.V2+ < Cr2+ < Mn2 < Fe2+ : paramagnetic behaviour
B.Ni2+ < Co2+ < Fe2+ < Mn2+ : ionic size
C.Co3+ < Fe3+ < Cr3+ < Sc3+ :stability in aqueous solution
D.Sc < Ti < Cr < Mn : number of oxidation states
Solution
V2+ = [Ar] 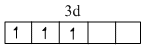
Cr2+ = [Ar]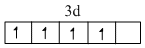
Mn2+ = [Ar]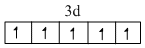
Fe2+ = [Ar]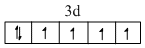
Correct order of Paramagnetic behaviour
Mn2+ > Fe2+ = Cr2+ > V2+

Cr2+ = [Ar]

Mn2+ = [Ar]

Fe2+ = [Ar]

Correct order of Paramagnetic behaviour
Mn2+ > Fe2+ = Cr2+ > V2+
Create a free account to view solution
View Solution FreeMore Electrostatics Questions
A metallic solid sphere is placed in a uniform electric field. The lines of force follow the path (s) shown in figure as...A charged water drop of radii 0.1 mm is under equilibrium in some electric field. The charge on the drop is equivalent t...Select the correct alternative :...Two identical small spheres carry charge of Q1 and Q2 with Q1>>Q2. The charges are d distance apart. The force the...Two charges + q and + q are r distant apart. The force between them is F. If one is fixed and other rotates in circle of...