ElectrostaticsHard
Question
The rate of a reaction doubles when its temperature changes from 300 K to 310 K. Activation energy of such a reaction will be
(R = 8.314 JK-1 mol-1 and log 2 = 0.301)
(R = 8.314 JK-1 mol-1 and log 2 = 0.301)
Options
A.53.6 kJ mol-1
B.48.6 kJ mol-1
C.58.5 kJ mol-1
D.60.5 kJ mol-1
Solution


∴


∴
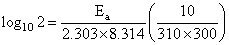
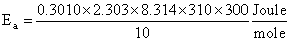
= 53.598 kJ/mol
Ea = 53.6 kJ/mol.
Create a free account to view solution
View Solution FreeMore Electrostatics Questions
Equipotential surfaces associated with an electric field which is increasing in magnitude along field which is increasin...The rupture of air medium occurs at E = 3 × 106 V/m. The maximum charge that can be given to a sphere of diameter 5...If the electric flux entering and leaving a closed surface are respectively of magnitude φ1. and φ2, then the ...The dielectric constant of a metal is : -...A conducting sphere of radius 10 cm is charged with 10 μC. another uncharged sphere of radius 20 cm is allowed to t...