Surface ChemistryHard
Question
For gaseous state, if most probable speed is denoted by C*, average speed by 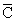 and mean square speed by C, then for a large number of molecules the ratios of these speeds are :
and mean square speed by C, then for a large number of molecules the ratios of these speeds are :
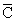 and mean square speed by C, then for a large number of molecules the ratios of these speeds are :
and mean square speed by C, then for a large number of molecules the ratios of these speeds are :Options
A.C* : 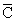 : C = 1.225 : 1.128 : 1
: C = 1.225 : 1.128 : 1
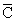 : C = 1.225 : 1.128 : 1
: C = 1.225 : 1.128 : 1B.C* : 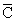 : C = 1.128 : 1.225 : 1
: C = 1.128 : 1.225 : 1
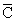 : C = 1.128 : 1.225 : 1
: C = 1.128 : 1.225 : 1C.C* : 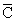 : C = 1 : 1.128 : 1.225
: C = 1 : 1.128 : 1.225
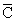 : C = 1 : 1.128 : 1.225
: C = 1 : 1.128 : 1.225D.C* : 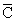 : C = 1 : 1.225 : 1.128
: C = 1 : 1.225 : 1.128
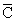 : C = 1 : 1.225 : 1.128
: C = 1 : 1.225 : 1.128Solution
Ump : Uavg : Urms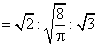
⇒ 1 : 1.128 : 1.224
∴ C* :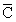 : C = 1 : 1.128 : 1.225
: C = 1 : 1.128 : 1.225
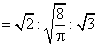
⇒ 1 : 1.128 : 1.224
∴ C* :
 : C = 1 : 1.128 : 1.225
: C = 1 : 1.128 : 1.225Create a free account to view solution
View Solution FreeMore Surface Chemistry Questions
Which of the following is a mismatch :...Which is not a purely surface phenomena :...Which one of the following statements is incorrect about enzyme catalysis?...Which one of the following statements is correct:...Colloidal solution of gold prepared by different methods of different colours because of :...