Surface ChemistryHard
Question
Graph between log x/m and log p is a straight line inclined at an angle of 45o. When pressure is 0.5 atm and ln k = 0.693, the amount of solute adsorbed per gm of adsorbent will be :
Options
A.1
B.1.5
C.0.25
D.2.5
Solution
log 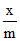 = log k +
= log k + 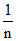 log P
log P
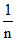 = tan 45o ln k = 0.69
= tan 45o ln k = 0.69
n = 1 k = 2
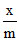 = 2 x (0.5)1
= 2 x (0.5)1
x = 1
n = 1 k = 2
x = 1
Create a free account to view solution
View Solution FreeMore Surface Chemistry Questions
Which of the following statements is false for catalyst?...Which among the following statements is false?...On addition of one ml solution of 10% NaCl to 10 ml gold sol in the presence of 0.0250 gm of starch the coagulation is j...The stabilisation of a lyophobic colloid is due to :...The size of a colloidal particle is :...