Surface ChemistryHard
Question
The pressure of the gas was found to decrease from 720 to 480 mm. When 5g of sample of activated charcoal was kept in a flask of one litre capacity maintained at 27oC. If the density of charcoal at 1.25 gm/mL. The volume of gas adsorbed per gm of charcoal at 480 mm of Hg is
Options
A.80.03 mL
B.32.20 mL
C.100.08 mL
D.None of these
Solution
V2 = 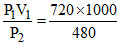 = 1500 ml
= 1500 ml
Volume of gas in the flask = 1000 - volume of charcoal = 1000 -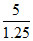 = 996 ml
= 996 ml
Total volume of gas adsorbe by charcoal = 1500 - 996 = 504 ml
Volume of gas adsorbe by per gram of charcoal =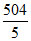 = 100.08 ml
= 100.08 ml
Volume of gas in the flask = 1000 - volume of charcoal = 1000 -
Total volume of gas adsorbe by charcoal = 1500 - 996 = 504 ml
Volume of gas adsorbe by per gram of charcoal =
Create a free account to view solution
View Solution FreeMore Surface Chemistry Questions
An emulsion is a colloidal solution consisting of...The colloidal system consisting of a liquid adsorbate in a solid adsorbent is termed as :...Which characteristic of adsorption is wrong?...Which of the following are multimolecular colloids ?...(i) At 298 K the volume of NH3 adsorbed by 1g of charcoal is higher than that of H2 under similar conditions.(ii) The mo...