Surface ChemistryHard
Question
Volume of N2 at NTP required to form a mono layer on the surface of iron catalyst is 8.15 ml/gram of the adsorbent. What will be the surface area of the adsorbent per gm if each nitrogen molecule occupies 16 ×10-22 m2.
Options
A.16 × 10-16 cm2
B.0.35 m2/g
C.39 m2/g
D.22400 cm2
Solution
The volume of N2 at STP required to cover the iron surface with monolayer = 8.15 ml gm-1
Area occupied by single molecule = 16 × 10-18 cm2
22400 ml of N2 at STP contains = NA molecule of N2
∴ 8.15 ..................... =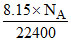 = 2.19 × 1020 molecule of N2
= 2.19 × 1020 molecule of N2
Area occupied by 2.19 × 1020 molecule of N2 = 2.19 × 1020 × 16 × 10-18 cm2 = 35.06 × 102 cm2
surface area of the iron adsorbed = 0.35 m2 gm-1
In short
A =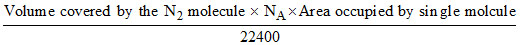
Area occupied by single molecule = 16 × 10-18 cm2
22400 ml of N2 at STP contains = NA molecule of N2
∴ 8.15 ..................... =
Area occupied by 2.19 × 1020 molecule of N2 = 2.19 × 1020 × 16 × 10-18 cm2 = 35.06 × 102 cm2
surface area of the iron adsorbed = 0.35 m2 gm-1
In short
A =
Create a free account to view solution
View Solution Free