Surface ChemistryHard
Question
Finely divided catalyst has greater surface area and has greater catalytic activity than the compact solid. If a total surface area of 6291456 cm2 is required for adsorption in a catalysed gaseous reaction, then how many splits should be made to a cube of exactly 1 cm in length to achieve required surface area.
(Given : One split of a cube gives eight cubes of same size)
(Given : One split of a cube gives eight cubes of same size)
Options
A.60
B.80
C.20
D.22
Solution
Total surface area of eight cubes = 8 × 6 × 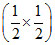
Apply the formula
Surface area on n split of a cube = 8n × 6 ×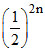
6291456 = 8n × 6 ×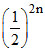
Apply the formula
Surface area on n split of a cube = 8n × 6 ×
6291456 = 8n × 6 ×
Create a free account to view solution
View Solution FreeMore Surface Chemistry Questions
Which of the following statement(s) regarding adsorption is/are not correct?...Which of the following are examples of aerosols?...Fe3+ ions coagulate blood. This shows that blood contains colloidal particles bearing...If dispersed phase is liquid and the dispersion medium is solid, the colloid is known as...On adding AgNO3 solution into KI solution, a negatively charged colloidal sol is obtained when they are in...