Solid StateHard
Question
In a hypothetical solid C atoms form CCP lattice with A atoms occupying all the Tetrahedral Voids and B atoms occupying all the octahedral voids.A and B atoms are of the appropriate size such that there is no distortion in the CCP lattice.Now if a plane is cut(as shown) then the cross section would like.
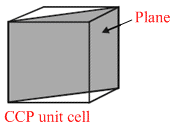

Options
A.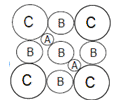

B.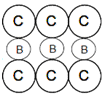

C.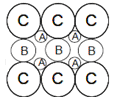

D.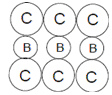

Solution
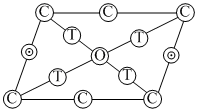
 → Octahedral void, at edge center & body center.
→ Octahedral void, at edge center & body center.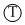 → Tetrahedral voids on body diagonal.
→ Tetrahedral voids on body diagonal.Create a free account to view solution
View Solution FreeMore Solid State Questions
Which statements is/are true about HCP and CCP lattice...The number of next nearest neighbours of Cs+ ion in CsCl crystal is...The distance between adjacent, oppositely charged ions in rubidium chloride is 3.285 A0; in potassium chloride is 3.139 ...An element crystallises in a ′bcc′ lattice. Nearest neighbours and next nearest neighbours of the elements a...In Na2O structure,...