Work, Power and EnergyHard
Question
The potential energy function for the force between two atoms in a diatomic molecule is approximately given by U(x) = 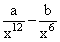 where a and b are constants and x is the distance between the atoms. If the dissociation energy of the molecule is D = [U(x = ∞) - Uat equilibrium], D is
where a and b are constants and x is the distance between the atoms. If the dissociation energy of the molecule is D = [U(x = ∞) - Uat equilibrium], D is
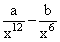 where a and b are constants and x is the distance between the atoms. If the dissociation energy of the molecule is D = [U(x = ∞) - Uat equilibrium], D is
where a and b are constants and x is the distance between the atoms. If the dissociation energy of the molecule is D = [U(x = ∞) - Uat equilibrium], D isOptions
A.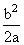
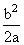
B.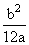
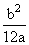
C.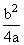
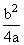
D.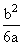
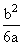
Solution
U(x) = 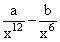
U(x = ∞) = 0
as,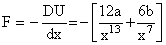
at equilibrium, F = 0
∴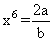
∴ Uat equilibrium =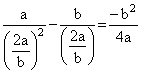
∴ D = [U(x = ∞) - Uat equilibrium] =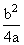
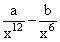
U(x = ∞) = 0
as,
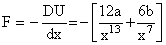
at equilibrium, F = 0
∴
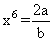
∴ Uat equilibrium =
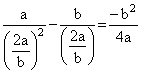
∴ D = [U(x = ∞) - Uat equilibrium] =
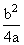
Create a free account to view solution
View Solution FreeMore Work, Power and Energy Questions
A stone of mass m is tied to a string of length l at one end and by holding second end it is whirled into a horizontal c...A block of mass 16 kg is moving on a frictionless horizontal surface with velocity 4m/s and comes to rest after pressing...Two masses of 1 g and 4 g are moving with equal kinetic energies. The ratio of the magnitudes of their momenta is...A ball falls from a height of 5m and strikes the roof of a lift. If at the time of collision, lift is moving in the upwa...A ball is dropped from height h on the ground level. If the coefficient of restitution is e then the height upto which t...