Solid StateHard
Question
For an Ionic solid of the general formula AB and coordination number 6, the value of the radius ratio will be
Options
A.less than 0.225
B.in between 0.225 and 0.414
C.between 0.414 and 0.732
D.greater than 0.732
Solution
Ionic solid having C.N. = 6 cation in octahedral holes.
Hence, 0.414 <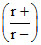 < 0.732
< 0.732
Hence, 0.414 <
Create a free account to view solution
View Solution FreeMore Solid State Questions
The volume of atoms present in a face-centred cubic unit cell of a metal (r is atomic radius) is:-...In the zinc blend structure (ZnS), S2− adopts CCP arrangement and Zn2+ occupies...If the positions of Na+ and Cl- are interchanged in NaCl, the crystal lattice with respect to Na+ and CI- is...A solid contains An+ and Bm− ions. The structure of solid is FCC for Bm− ions and An+ ions are present in one-fourth of ...F-centers are...