Solid StateHard
Question
In a cubic structure of compound which is made from X and Y, where X atoms are at the corners of the cube and Y at the face centers of the cube. The molecular formula of compound is
Options
A.X2Y
B.X3Y
C.XY2
D.XY3
Solution
for X, 8 × 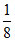 = 1
= 1
for Y, 6 ×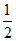 = 3
= 3
so, XY3
for Y, 6 ×
so, XY3
Create a free account to view solution
View Solution FreeMore Solid State Questions
Aluminium crystallizes in cubic system with unit cell edge length equal to 4.0 Å. If its density is $\frac{45}{16}$g/cm3...In the closest packing of atoms,...A crystal of NaCl, which has sodium ion and chloride ion missing from the lattice point, is said to exhibit...In the crystal lattice of diamond, carbon atoms adopt :...A solid contains An+ and Bm− ions. The structure of solid is FCC for Bm− ions and An+ ions are present in one-fourth of ...