Momentum and CollisionHard
Question
Which one of the following molecules is expected to exhibit diamagnetic behaviour ?
Options
A.C2
B.N2
C.O2
D.S2
Solution
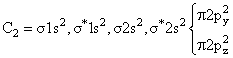 (Diamagnetic)
(Diamagnetic) 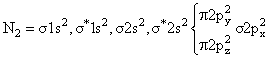 (Diamagnetic)
(Diamagnetic)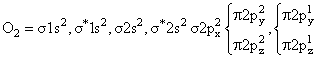 (Paramagnatic)
(Paramagnatic)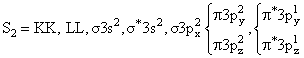 (Paramagnetic)
(Paramagnetic)C2 & N2 both are diamegnatic actully, but according to the language of question,expected to be diamegnatic.
If we consider energy leval diagram of C2 and N2 similar to O2 then C2 become paramagnetic & N2 remain diamegnatic so best answer of Question is N2.
Create a free account to view solution
View Solution FreeMore Momentum and Collision Questions
A ′T′ shaped object with dimensions shown in the figure, is lying on a smooth floor. A force F is applied at...Find the x coordinate of the centre of mass of the bricks shown in figure :-...The lower surface of a cube is fixed. On its upper surface, force is applied at an angle of 30o from its surface. The ch...A rain drop of radius 0.3 mm has a terminal velocity in air 1 m/s. The viscosity of air is 18 × 10-5 poise. The vis...A crown made of gold and copper weights 210 g in air and 198 g in water. The weight of gold in crown is : -[Given : Dens...