Surface ChemistryHard
Question
When a graph is plotted between log x/m and log p, it is straight line with an angle 45o and intercept 0.3010 on y-axis. If initial pressure is 0.3 atm, what will be the amount of gas adsorbed per gm of adsorbent :
Options
A.0.4
B.0.6
C.0.8
D.0.1
Solution
log 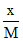 = log k +
= log k + 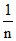 log P
log P
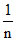 = tan 45o and log k = 0.3010
= tan 45o and log k = 0.3010
n = 1 and k = 2
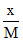 = 2 × (0.3)1
= 2 × (0.3)1
x = 0.6
x = 0.6
Create a free account to view solution
View Solution FreeMore Surface Chemistry Questions
Blood may be purified by...Charge on above colloidal structure is :-...On adding AgNO3 solution into KI solution, a negatively charged colloidal sol is obtained when they are in...Which plot is the adsorption isobar for chemisorption? (b)...Which one of the following statements regarding photochemical smog is not correct?...