Periodic TableHard
Question
The correct sequence which shows decreasing order of the ionic radii of the elements is
Options
A.Al3+ > Mg2+ > Na+ > F- > O2-
B.Na+ > Mg2+ > Al3+ > O2- > F-
C.Na+ > F- > Mg2+ > O2- > Al3+
D.O2- > F- > Na+ > Mg2+ > Al3+
Solution
For isoelectronic species higher the 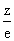 ratio , smaller the ionic radius
ratio , smaller the ionic radius
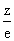 for O2 =
for O2 = 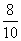 = 0.8
= 0.8
F- =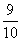 = 0.9
= 0.9
Na+ =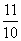 = 1.1
= 1.1
Mg2+ =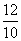 = 1.2
= 1.2
Al3+ =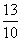 = 1.3
= 1.3
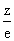 ratio , smaller the ionic radius
ratio , smaller the ionic radius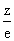 for O2 =
for O2 = 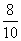 = 0.8
= 0.8F- =
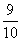 = 0.9
= 0.9Na+ =
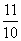 = 1.1
= 1.1Mg2+ =
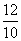 = 1.2
= 1.2Al3+ =
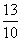 = 1.3
= 1.3Create a free account to view solution
View Solution FreeMore Periodic Table Questions
In which of the following options the order of arrangement does not agree with the variation of propertyindicated agains...The statements that is / are true for the long form of the periodle is / are :...Which one of the following sets of ions represents a collection of isoelectronic species?...The incorrect statement among the following is :...which of the following pairs has the same size ?...