Redox and Equivalent ConceptHard
Question
29.5 mg of an organic compound containing nitrogen was digested according to Kjeldahl′s method and the evolved ammonia was absorbed in 20 mL of 0.1 M HCl solution. The excess of the acid required 15 mL of 0.1 M NaOH solution for complete neutralization. The percentage of nitrogen in the compound is
Options
A.59.0
B.47.4
C.23.7
D.29.5
Solution
Moles of HCl reacting with
ammonia = (moles of HCl absorbed ) - (moles of NaOH solution required)
= (20 × 0.1 × 10-3) - (15 × 0.1 × 10-3)
= moles of NH3 evolved.
= moles of nitrogen in organic compound]
∴ wt. of nitrogen in org. comp = 0.5 × 10-3 × 14
= 7 × 10-3 g
% wt =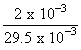 = 23.7 %
= 23.7 %
ammonia = (moles of HCl absorbed ) - (moles of NaOH solution required)
= (20 × 0.1 × 10-3) - (15 × 0.1 × 10-3)
= moles of NH3 evolved.
= moles of nitrogen in organic compound]
∴ wt. of nitrogen in org. comp = 0.5 × 10-3 × 14
= 7 × 10-3 g
% wt =
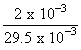 = 23.7 %
= 23.7 %Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
Ge (II) compounds are powerful reducing agents whereas Pb (IV)compounds are strongoxidants. It can be due to....Which of the following reaction is an example of comproportionation reaction?...In the following reaction (unbalanced), equivalent weight of As2S3 is related to its molecular weight, M, byAs2S3 + H+ +...The equivalent weight of ozone behaving as an oxidizing agent is...In basic medium, Cl2 disproportionates into Cl− and ClOx−. If there is loss and gain of one mole of electron per mole of...