Nuclear Physics and RadioactivityHard
Question
Reaction by which Benzaldehyde cannot be prepared :-
Options
A.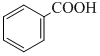 + Zn/Hg and conc. HCl
+ Zn/Hg and conc. HCl
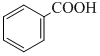 + Zn/Hg and conc. HCl
+ Zn/Hg and conc. HClB.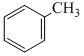 + CrO2Cl2 in CS2 followed by H3O+
+ CrO2Cl2 in CS2 followed by H3O+
 + CrO2Cl2 in CS2 followed by H3O+
+ CrO2Cl2 in CS2 followed by H3O+C.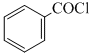 + H2 in presence of ( Pd+BaSO4)
+ H2 in presence of ( Pd+BaSO4)
 + H2 in presence of ( Pd+BaSO4)
+ H2 in presence of ( Pd+BaSO4)D.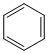 + CO + HCl in presence of anhydrous AlCl3
+ CO + HCl in presence of anhydrous AlCl3
 + CO + HCl in presence of anhydrous AlCl3
+ CO + HCl in presence of anhydrous AlCl3 Solution
In presence of Zn - Hg and conc. HCl reduction is useful specially for aldehyde and ketone but carboxylic group remains uneffected








Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
Binding energy per nucleon of deutron is 1.112 MeV and binding energy per nucleon of an α particle is 7.07 MeV, the...If radius of nucleus is estimated to be 3.6 Fermi then the radius nucleus be nearly...The masses of two isobars $^{64}_{29}\text{Cu}$ and $^{64}_{30}\text{Zn}$ are $63.9298 \text{ u}$ and $63.9292 \text{ u}...Outside the nucelus :...In a sample, ratio of p31 and p32 is 1 : 2. P31 is stable but p32 is radioactive and decays in S32 with half life T. Aft...